15+ Years of Biosimilar Experience in Europe: Omnitrope Case Study
Biosimilar competition can play a crucial role in the overall economic sustainability of the healthcare system. Biosimilars hold the potential to improve patient access to biologic therapies, balance spending, and unlock savings for innovation. IQVIA Institute has previously published reports on biosimilar sustainability (Advancing Biosimilar Sustainability, 2018, and Spotlight on Biosimilars, 2021).
This new report continues the conversation by assessing the status of biosimilar use in selected European countries and, in particular, by analyzing the value provided by the first biosimilar launched in Europe — Omnitrope®. This utilization of Omnitrope and the savings associated with it are assessed using proprietary IQVIA data for selected European countries. With more than 15 years since its approval, Omnitrope’s value to the overall health system is important to understand and can provide lessons for ensuring that future biosimilars are optimally utilized.
Please submit the below form to download
Key findings:
- Over the past 15+ years, several studies have been conducted that show Omnitrope® to be highly similar to Genotropin, the reference product in pharmacokinetic and pharmacodynamic safety and efficacy profiles, and comparable in quality, safety, and efficacy.
- In these 15+ years, Omnitrope has also witnessed access and uptake in most European countries and has a 30% share of overall somatropin usage; however, its uptake trajectory differs from that of more recent biosimilar products.
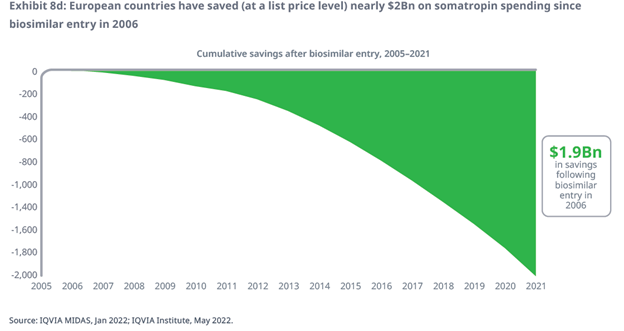
- Based on analysis of IQVIA MIDAS data, the entry of Omnitrope (somatropin) has led to more than $1.9 billion in savings at a list price level across selected European countries between 2006 and 2021, driven by its reduced cost and the increased competitive pressure on reference medicines.
- The level of savings varies across countries, considering biosimilar uptake, competitive response and regulations are different.
Other findings:


- There has been a gradual increase in the Omnitrope share of the somatropin market in terms of volume as more countries have provided access to the biosimilar and there has been greater acceptance of it across all countries; the percentage share increased from 13% in 2012 to 30% in 2022.
- Overall, there has been an increase of 101% in annual somatropin volume use since 2006 while the overall sales have only increased by 34%.
- Across the countries, Omnitrope has had greater than 300,000 patient years of usage over the 15 years, with the highest use taking place in France, Spain, Germany, and Poland.
- The total cumulative savings associated with Omnitrope across all countries since 2006 is estimated to be $1.9 billion at a list price level (approximately 14% of the overall somatropin spending in the same period) with the highest savings taking place in France, Spain, the UK, and Italy.

Copyright © 2023 IQVIA Holdings Inc. and its affiliates. All rights reserved. Privacy Statement
